If the oxygen of the anomeric carbon - initially carbonyl group - of a sugar is not bound to any other structure, sugar is a reducing sugar, that is, it can donate electrons to another molecule. Many carbohydrates are able to change spontaneously between the two configurations by a rotation. The result also, of this rotation, are the two configurations previously mentioned like alpha and beta. In most cases, the anomeric carbons can be identified as seen above, when a carbon bonded to two single bond carbon atoms is located. The best examples of anomeric carbons are found in monosaccharides such as glucose, in which a rotation occurs. It is known, therefore, that the electronegative element which is in the anomeric position will be located in the axial location. This also happens in carbohydrates, acyclic systems and saturated heterocycles. Properties of anomeric carbonsĪnomeric carbons possess the property known as anomeric effect, which is the preference of the electronegative element to be in the axial position as opposed to the equatorial.Īnomeric carbon has a property known as the anomeric effect, which is the preference for an electronegative to be in an axial orientation as opposed to the equatorial orientation. 
The formation of rings of the linear sugars which are less stable, is through the union of an aldehyde (-C = OH) and an alcohol group (-OH), giving rise to the hemiacetal bond. However, when hydrogen remains bound and oxygen is not bound to the COC form, it is called the hemiacetal group because the OH group can close and open to any shape in a mixture of these molecules, so there are some alpha and Other beta. Once this happens, the anomeric carbon is fixed either in the alpha or beta position. 
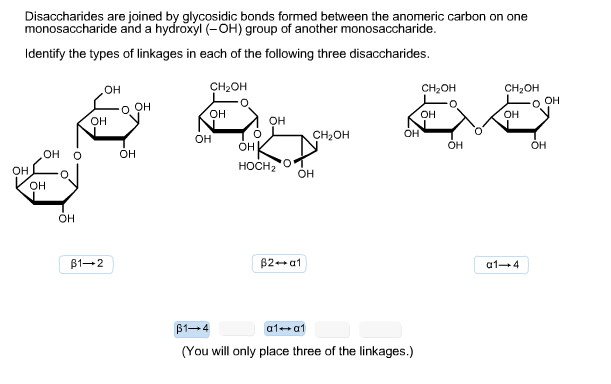
When two monosaccharides are bound, they do so through an acetal group (C-O-C-O). It is common for both cyclic forms (alpha and beta) to be in equilibrium in aqueous solution, and to convert spontaneously into one another in a process called 'mutarotation'. Thus, for example, glycogen is synthesized by alpha-D-glucanopyranose, while cellulose is synthesized by beta-D-glucopyranose. The importance of this is that there are enzymes that are able to distinguish between both structures and have preference for one of them. 2 are both glucose, but anomers with respect to each other. On the right, alcohol group down, is alpha, and the molecule is known as alpha-D-glucopyranose, in the figure on the right, the OH group is up, and is known as beta- D-glucopyranose. This will be called a when it is down in the equatorial, or axial, form And b, when it is upward in both the axial and equatorial positions.įig. In the cyclic form, the anomeric carbon can be found next to the oxygen atom in the pyranose and furanose rings, but at the opposite site of the carbon bearing the linear group CH 2 OR.īeing a stereocenter the anomeric carbon, an important characteristic, is the direction that acquires the group alcohol (-OH) attached to that carbon, since depending on its direction is denominated a or b. 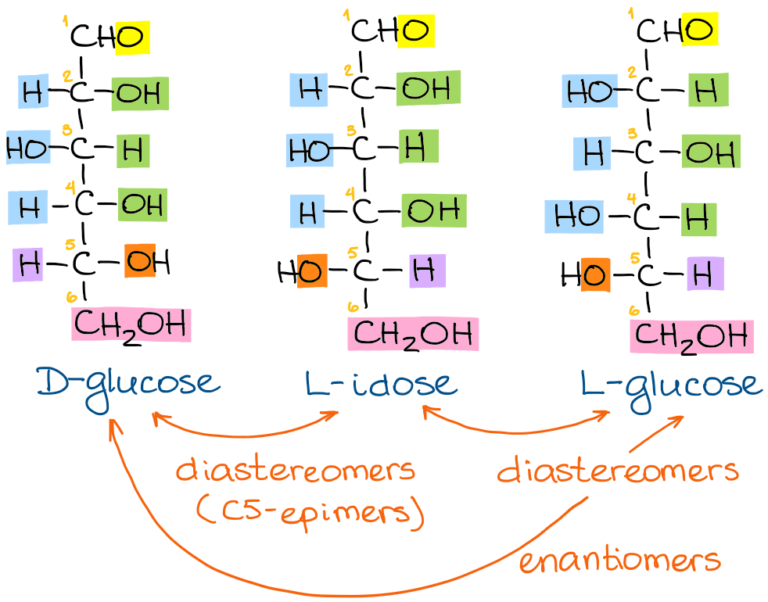
Thus, in a cyclic carbohydrate, the anomeric carbon will be (UCLA, 2015) which was carbonyl (CH 2 O) in the acyclic or linear form. One will be attached to the CH group 2 OH, and the other carbon that is not bound to that group will be the anomeric carbon. Once the cycle has been formed, to locate the anomeric carbon (Chang, n.d.), first locate the oxygen inside the ring, and then look at the carbon on both sides of the ring. Position of anomeric carbon inside the ring The new stereocenter is then called anomeric carbon. The diastereoisomers formed differ in the position of a group, in the new stereocenter formed. When a stereocenter is formed in the cyclization, two new"diastereoisomers"also occur, which are a class of stereoisomers, which are mirror images of each other, but not superimposable, that is to say, they are not"enantiomers". By"stereocenter", carbon is understood to have four different substituents, also called"chiral carbon". Usually occurs in sugars, which are simply simple carbon hydrated compounds, where the most common are: glucose, fructose, lactose and galactose.ĭuring cyclization, the carbonyl moiety that formed part of that linear structure is transformed into a new stereocenter. Therefore, most are in cyclic form, or also referred to as ring, wherein the functional group either aldehyde or ketone, has reacted with an alcohol of the same sugar. Less than one percent of each of the monosaccharides (Champe, Harvey, & Ferrier, 2005) with five or more carbons exists in the form of a chain, that is, they are linear or acyclic.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
